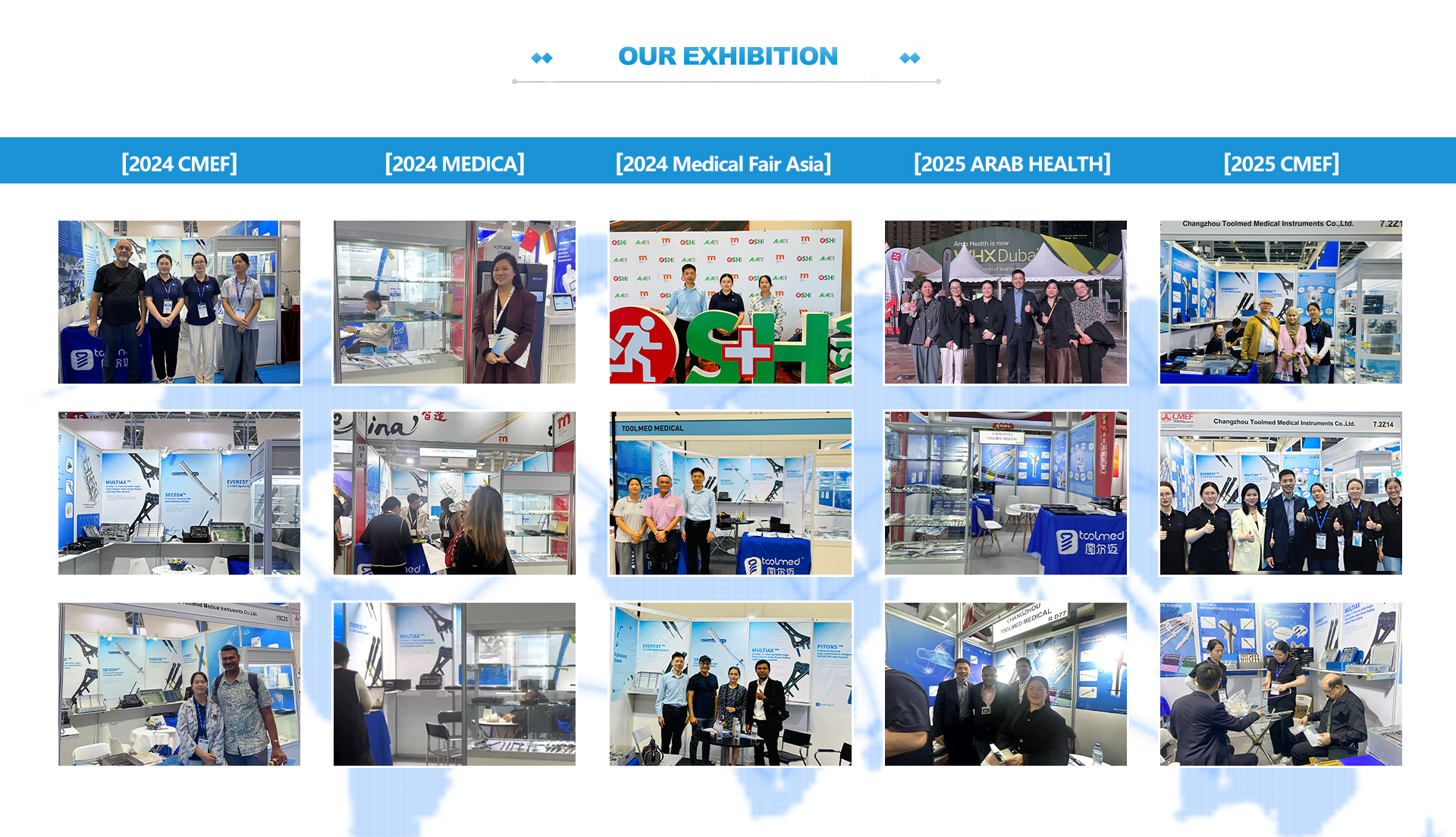
Views: 0 Author: Site Editor Publish Time: 2026-02-26 Origin: Site
Arthroscopic surgery is defined by precision, efficiency, and minimal disruption to surrounding tissues. While attention is often focused on optics, shavers, and powered instruments, access devices such as cannulas play a critical but sometimes underestimated role in procedural success. An inadequately designed cannula can increase soft tissue trauma, compromise fluid management, or slow instrument exchange, ultimately affecting both surgical efficiency and patient outcomes.
The Arthroscopic Silicone Cannula has emerged as a specialized solution addressing these challenges. Unlike rigid plastic or metal alternatives, silicone-based cannulas prioritize flexibility, soft tissue compatibility, and dynamic sealing performance. Their role extends beyond simple portal maintenance—they actively contribute to a smoother surgical workflow and a more controlled arthroscopic environment.
This article examines the design rationale, material characteristics, surgical applications, and long-term clinical value of Arthroscopic Silicone Cannulas within modern minimally invasive orthopedic surgery.
Cannulas serve as controlled access pathways between the external environment and the joint space. They protect surrounding soft tissues from repeated instrument insertion and help maintain consistent portal geometry throughout the procedure.
In high-frequency instrument exchanges—common in shoulder, knee, and hip arthroscopy—portal stability directly affects surgical rhythm and operative time.
Arthroscopy relies heavily on pressurized fluid to maintain joint distension and clear visualization. Cannula design influences fluid inflow, outflow, and leakage around the portal site. Even minor inefficiencies can result in fluid extravasation, tissue swelling, and compromised visualization.
Medical-grade silicone is selected for arthroscopic cannulas due to a combination of mechanical and biological characteristics:
These properties allow the cannula to conform dynamically to soft tissue and instrument contours rather than resisting them.
Rigid plastic or metal cannulas provide dimensional stability but lack adaptability. In contrast, silicone cannulas absorb minor angular deviations during instrument insertion, reducing localized stress on portal tissues.
This distinction becomes particularly important in anatomically constrained joints such as the shoulder, where portal angles frequently change during the procedure.
A well-designed silicone cannula must strike a balance between softness and shape retention. Excessive flexibility can cause lumen collapse, while insufficient compliance negates the benefits of silicone.
Modern Arthroscopic Silicone Cannulas are engineered to maintain a consistent internal diameter while allowing controlled deformation under load.
Many silicone cannulas incorporate valve or diaphragm features that adapt around instruments. These seals reduce fluid leakage during insertion and withdrawal, helping maintain intra-articular pressure.
Key functional goals of sealing design include:
Smooth instrument exchange is one of the most tangible benefits reported by surgeons using silicone cannulas. The compliant material reduces friction and resistance, allowing instruments to pass without catching or damaging portal edges.
This efficiency becomes particularly valuable in procedures requiring frequent transitions between probes, shavers, graspers, and suture passers.
Repeated instrument manipulation can traumatize skin, fascia, and capsular tissue. Silicone cannulas act as a protective interface, reducing shear forces and lowering the risk of postoperative pain or portal site complications.
In shoulder procedures, silicone cannulas support dynamic portal angles and accommodate a wide range of instrument diameters. Their flexibility is particularly advantageous when working in the subacromial space or around the glenoid.
In knee arthroscopy, cannulas contribute to fluid containment and efficient instrument handling. In hip arthroscopy—where portals traverse thicker soft tissue layers—silicone cannulas help reduce access resistance and tissue strain.
Unlike rigid polymer injection molding, silicone component manufacturing requires careful control of curing, wall thickness, and elasticity. Consistency is critical to ensure predictable sealing behavior and lumen stability.
Precision-controlled silicone molding processes support:
For distributors and OEM partners, traceability of silicone raw materials and process validation is essential. Reliable quality systems ensure that each cannula performs consistently under surgical conditions.

Medical-grade silicone is compatible with standard sterilization techniques, including ethylene oxide and gamma irradiation. Its structural integrity and elasticity remain stable when processed correctly.
Silicone’s long-standing history in implantable and non-implantable medical devices supports its safety profile. Cannulas manufactured under controlled quality systems meet regulatory expectations for biocompatibility and performance.
From a surgeon’s standpoint, Arthroscopic Silicone Cannulas provide:
These benefits contribute directly to operative efficiency and procedural confidence.
For distributors and OEM partners, value is defined by:
Silicone cannulas function as essential system components rather than disposable accessories.
The TOOLMED’s Arthroscopic Silicone Cannula exemplifies how thoughtful material selection and design refinement can meaningfully improve minimally invasive surgical workflows. By prioritizing flexibility, tissue compatibility, and fluid control, silicone cannulas address real challenges encountered in daily arthroscopic practice.
Their value lies not in technological novelty, but in dependable performance where precision, efficiency, and patient comfort intersect. As arthroscopic techniques continue to evolve, access devices like silicone cannulas will remain foundational to successful outcomes.
JBJS Orthopaedic Trauma: Which Gold Standards Have Been Overturned in the Past 7 Years?
Surgical Site Infection (SSI) in Orthopedic Wounds: Prevention And Treatment Consensus
Bolt Locking vs Cap Locking in Orthopedic Locking Plates: Key Differences Explained
Top 15 Orthopedic Manufacturers in America for Distal Humerus Locking Plates (2026)
2.0mm Locking Maxillofacial Plate System: Precision Fixation within the CMF Surgical Framework
Links
Contact Us